Fadhil Fabiano Hendis
Contents
- 1 INTRODUCTION
- 2 MR. DAI CCIT House Visit
- 3 CHAT SESSION WITH CHATGPT
- 4 CLASS APOLOGY ON 24/02/2023
- 5 CLASS SUMMARY 24/02/2023 ICE Modeling on OpenModelica
- 6 CLASS SUMMARY 03/03/2023 Pyrolysis
- 7 CLASS SUMMARY 07/03/2023 Desalination
- 8 QUIZ 02 28/03/2023
- 9 Hands-on PROJECT
- 10 Hands-on PROJECT PROGRAMMING
- 11 CLASS SUMMARY 02/05/2023 Discussion with Prof. Adi
INTRODUCTION
My name is Fadhil Fabiano Hendis NPM 2006489110, I've been referred to as Fadhil or Bian. I am 20 years old, I was born on 25 February 2003 in south Jakarta and I’m currently in my sixth semester of study at Universitas Indonesia.
I have plenty interest in life, but the one I'd like to share is photography. Having a small memory kept in the form of a picture appeals to me, and I believe that one photograph may hold many story for other people. Even if I'm still a beginner, I want to learn more!
I appreciate you taking the time to read this brief essay about me. Thank you.
MR. DAI CCIT House Visit
On Friday, my friends and I visited CCIT and were amazed not only by its exterior, but also by the wide range of content inside. What impressed me the most was the abundance of Azolla pools. Azolla plants have a high crude protein content, ranging from 23% to 30%, and a low lignin content, making them easily digestible for livestock. Thus, Azolla plants are an excellent source of animal feed, especially for ducks, chickens, goats, cows, and rabbits.
Mr. Tanwir and Ms. Illa also explained a machine that uses Azolla and animal feces as its fuel source. These materials are heated up to 300-400 degrees Celsius using flue gases from a generator to power the machine. The gases then flow up from the pyrolysis tank to the condenser. This process is considered environmentally friendly since it uses green energy from Azolla and animal feces. Furthermore, Mr. DAI has a handsome goat named Saun.
During the visit, we learned that Azolla plants are not only a good source of animal feed but also have several environmental benefits. Azolla is a natural biofertilizer, which can be used to enrich soil and reduce the need for chemical fertilizers. Moreover, Azolla is known for its ability to fix atmospheric nitrogen, which makes it a potential alternative to synthetic nitrogen fertilizers that contribute to greenhouse gas emissions. We were amazed to learn about the potential of Azolla plants in promoting sustainable agriculture and reducing the environmental impact of animal husbandry. The visit to CCIT was not only educational but also inspiring, and we left with a newfound appreciation for the potential of Azolla plants as a sustainable solution for feeding the world's growing population.
CHAT SESSION WITH CHATGPT
Fadhil : What’s the correlation between battery factory, sugar factory and fuel?
ChatGPT : The correlation between a battery factory, sugar factory, and fuel is related to the production and use of ethanol, which is a type of alcohol that can be used as a fuel or as a component of battery electrolytes. Sugar factories are known for producing sugar from sugar cane or sugar beets. However, during the process of extracting sugar from these plants, a byproduct known as molasses is produced. Molasses is a thick, dark syrup that is rich in sugars, minerals, and other nutrients. Molasses can be used as a feedstock for the production of ethanol, which is typically produced through a process of fermentation and distillation. In some cases, sugar factories themselves may operate ethanol plants as a way to generate additional revenue from their molasses byproduct. Ethanol can be used as a fuel for vehicles, either on its own or blended with gasoline. It is also used as a component of battery electrolytes in some types of batteries, such as those used in electric vehicles. Ethanol is particularly useful in battery electrolytes because it is a highly polar solvent that can dissolve a variety of salts, allowing for the transfer of charged ions between the battery's electrodes. In summary, the correlation between a battery factory, sugar factory, and fuel is that sugar factories can produce molasses, which can be used as a feedstock for ethanol production. Ethanol can then be used as a fuel or as a component of battery electrolytes, which are used in batteries produced by battery factories. This connection highlights the interrelatedness of various industries in the production and use of different types of energy and materials.
Fadhil : Can sugarcane be the main source of ethanol?
ChatGPT : Sugarcane is one of the most important sources of ethanol today and it has the potential to be a major source of biofuel in the future. Sugarcane is an ideal source of ethanol because it is a high-yielding and energy-efficient crop, meaning that it produces a large amount of energy in relation to the amount of energy required to grow and process it. Additionally, sugarcane is relatively easy to grow and has a wide range of uses beyond ethanol production, such as producing sugar, molasses, and other products.
Fadhil : Can you show me the literature regarding "sugarcane as a source of ethanol" ?
ChatGPT : Certainly! Here are some references that provide more information about the use of sugarcane as a source of ethanol:
Ferreira-Leitão, V., Rodrigues, J., & Maciel Filho, R. (2016). A review on ethanol production from sugarcane bagasse: Chemical and technological perspectives. Chemical Engineering Transactions, 52, 1-22. https://doi.org/10.3303/CET1652001
Goldemberg, J., Coelho, S. T., & Guardabassi, P. (2004). The sustainability of ethanol production from sugarcane. Energy Policy, 32(6), 677-687. https://doi.org/10.1016/S0301-4215(03)00121-5
Macedo, I. C., Seabra, J. E. A., & Silva, J. E. A. R. (2008). Greenhouse gases emissions in the production and use of ethanol from sugarcane in Brazil: The 2005/2006 averages and a prediction for 2020. Biomass and Bioenergy, 32(7), 582-595. https://doi.org/10.1016/j.biombioe.2007.12.006
Rajendran, K., Rajoli, S. R., & Taherzadeh, M. J. (2016). Current status and future prospects of bioethanol production from sugarcane and cassava wastes in Thailand. Bioresource Technology Reports, 1, 50-56. https://doi.org/10.1016/j.biteb.2016.02.004
Rosillo-Calle, F., & Cortez, L. A. B. (2017). Sugarcane ethanol: Contributions to climate change mitigation and the environment. In S. E. Jørgensen, F. Rosillo-Calle, & J. Fargione (Eds.), Biofuels for a more sustainable future: An assessment of the sustainability of biofuels using selected sustainability indicators (pp. 49-76). Academic Press. https://doi.org/10.1016/B978-0-12-804195-9.00003-6
These references cover a range of topics related to sugarcane ethanol production and use, including the technological and chemical aspects of production, environmental impacts, and the current status and future prospects of bioethanol production from sugarcane wastes in different regions.
Fadhil : How did you know sugarcane is an ideal source for ethanol?
ChatGPT : As an AI language model, I have been trained on vast amounts of data and information. The statement that sugarcane is an ideal source for ethanol is based on scientific research and industry practice, which has been documented in academic papers, reports, and other sources. Sugarcane is a highly productive crop that is grown in tropical and subtropical regions around the world, particularly in countries such as Brazil, India, and Thailand. It is rich in fermentable sugars, which can be converted into ethanol through a relatively simple process. Additionally, sugarcane has a high energy yield, meaning that it produces more energy than is required to grow and process it. Research has also shown that sugarcane ethanol has a lower carbon footprint than gasoline and other fossil fuels, making it an attractive option for reducing greenhouse gas emissions and mitigating climate change. Overall, the suitability of sugarcane as a source of ethanol is based on a combination of factors, including its productivity, energy efficiency, and sustainability.
Fadhil : Are you sure about your data?
ChatGPT : As an AI language model, I strive to provide accurate information based on the data and information available to me. However, as with any source of information, there is always the potential for errors or inaccuracies. If you have any specific concerns or questions about the data I have presented, please feel free to let me know and I can try to provide further clarification or sources to support my statements.
SUMMARY
What I can think of is that AI can only be used as a supporting tool, never as the "main" one. due to the alleged uncertainly or unsureness of AI. When an artificial intelligence system is unable to confidently respond to a question or task, this is referred to as AI unsureness. Numerous factors, such as insufficient or conflicting data, ambiguous or unclear instructions, or restrictions in the algorithms or models the system is using, can cause this to happen.
AI systems are frequently designed to provide a level of certainty or confidence in their responses, such as by providing a probability score or confidence interval. However, in some cases, the system may be unable to provide a confident response and must instead indicate its uncertainty.
Users who are expecting a definitive answer or response may find this difficult, as uncertainty can lead to confusion or frustration. However, it is critical to recognize that uncertainty is a natural part of any decision-making process, whether performed by a human or an AI system.
To address AI uncertainty, researchers and developers are investigating new techniques and algorithms, such as probabilistic reasoning, fuzzy logic, and Bayesian networks, that can assist AI systems in dealing with ambiguity and uncertainty. These approaches aim to provide more robust and reliable AI systems capable of dealing with complex and uncertain situations.
Butvinik, D. (n.d.). Uncertainty Quantification in Artificial Intelligence-based Systems - KDnuggets. KDnuggets. Retrieved February 24, 2023, from https://www.kdnuggets.com/uncertainty-quantification-in-artificial-intelligence-based-systems.html
CLASS APOLOGY ON 24/02/2023
I regret what I did and apologize to Mr. DAI and Bang Tanwir for my lateness to class on Friday, February 24, 2023. I hope to change my attitude. Thank you Mr. DAI and Bang Tanwir
CLASS SUMMARY 24/02/2023 ICE Modeling on OpenModelica
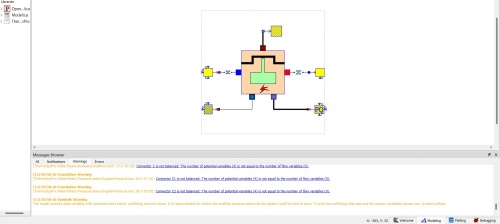
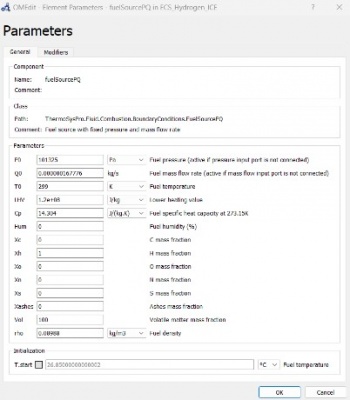
We were given a tutorial today with Bang Tanwir on OpenModelica for ICE. An internal combustion engine (ICE) burns fuel and air to produce power and fuel gases. Refrigerant cooling is also required for ICE. We can import the library because it is available in OpenModelica and contains the ICE, the library called ThermoSysPro.
These are modeling scheme on OpenModelica regarding Internal Combustion Engine.
These are the results from the simulation that I want to share. The following values are included: active power, fuel power available, engine electrical power, engine mechanical power, and power of thermal losses and cooling of the engine.
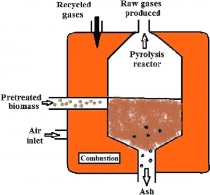
CLASS SUMMARY 03/03/2023 Pyrolysis
Class session is being led by two teaching assistants, with Professor Adi join in at the end. Their lecture revolves around the intricate science of combustion, exploring both thermochemical and biochemical processes, as well as various product-driven methods, such as gasification, liquid, and combustion. In particular, they delve into pyrolysis, an oxygen-starved combustion that requires biomass to be dried and cut prior to ignition for optimal energy output. To achieve the desired results, pyrolysis necessitates constant heat or the presence of a catalyst. One of its key benefits is the versatility of its end products, which can range from gases to liquids.
The end products of pyrolysis can be quite versatile, and they depend on various factors such as the type of biomass used, the temperature at which the pyrolysis occurs, and the method used to capture and store the end products.
Important product of pyrolysis is biochar, which is a carbon-rich solid material that is similar to charcoal. Biochar can be used as a soil amendment to improve soil quality, reduce greenhouse gas emissions, and sequester carbon in the soil.
Overall, the versatility of pyrolysis end products makes it an attractive technology for the conversion of biomass into a range of valuable products, including renewable energy, soil amendments, and specialty chemicals.
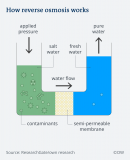
CLASS SUMMARY 07/03/2023 Desalination
Desalination is a method used to remove minerals from saltwater. Desalination more generally refers to the process of removing salts and minerals from a target substance, such as soil, which is problematic for agriculture. Saltwater can be desalinated to make it drinkable by boiling the water, catching the steam, and discarding the salt. Optionally, you could force the water through filters that capture the salt while letting the liquid through. Although the latter is the more contemporary procedure, both take a lot of energy. Let's talk about the second option, graphene can be chosen as a material for the desalination filter. Graphene is a carbon allotrope composed of a single sheet of atoms organized in a hexagonal lattice nanostructure. The name is taken from graphite, reflecting the abundance of double bonds in the carbon allotrope found in graphite. The use of graphene have outstanding filtering performance and mechanical qualities, but it is still hard to produced.
Biomass is material derived from recently living creatures and used in the creation of bioenergy. Examples include wood, wood byproducts, energy crops, agricultural waste, and organic waste from commercial and residential sources. The main biomass energy source nowadays is wood and wood waste. The light-weight, black residue that is left over after biomass is pyrolyzed and is composed of carbon and ashes is known as biochar.
In summary, Desalination is a method of removing minerals from saltwater, which can be achieved by boiling the water and discarding the salt or using filters to capture the salt while letting the liquid through. Graphene is a material that can be used for desalination filters due to its outstanding filtering performance and mechanical qualities. However, it is still challenging to produce. Biomass is a material derived from recently living creatures that can be used for bioenergy, and examples include wood, energy crops, agricultural waste, and organic waste. Wood and wood waste are currently the main biomass energy sources. Biochar is the black residue left over after biomass is pyrolyzed and is composed of carbon and ashes.
PowerPoints Presentation
https://docs.google.com/presentation/d/1cVkoBv4dX9qNwmZ2o1ZwEI4AJeV9l4BAAq0e_CRN7p8/edit?usp=sharing
QUIZ 02 28/03/2023
Hands-on PROJECT
YOUTUBE LINK
INTRO
We were given a joint project by Mr. DAI and Prof. Adi so that we could learn more about the ECS 02 topic. This project will examine the power output produced by a hydro fuel-powered generator with different parameters of plate type, thickness, and dimension. It is based on Rasendriya's project on gathering hydro fuel.
Code Using Python - https://colab.research.google.com/drive/1mAKCXZdlIy7-DY3iQlseKfdKfyE0pyLk
Personal Input/Suggestion
I have an input on how to improve the electrolysis bubble formation and collection in order to increase the yield of hydrogen gas.
Firstly, Increase the surface area of the electrodes. Electrolysis reactions occur at the surface of the electrodes, so increasing the surface area of the electrodes can increase the amount of hydrogen bubbles that are generated during electrolysis. This can be achieved by using electrodes with a larger surface area, or by increasing the number of electrodes in the electrolysis cell.
Second, Increase the voltage. Electrolysis requires energy input in the form of an applied voltage, so increasing the voltage applied to the electrodes can increase the rate of hydrogen bubble formation. However, care should be taken to avoid overvoltage, which can result in the production of unwanted byproducts or damage to the electrodes.
Third, Use a catalyst. A catalyst is a substance that can increase the rate of a chemical reaction without being consumed in the process. Using a catalyst on the surface of the electrodes can increase the rate of hydrogen bubble formation, and can also make the bubbles smaller and easier to collect. Catalysts commonly used for electrolysis of water include platinum, iridium, and ruthenium.
Fourth, Improve electrolyte circulation. Electrolyte circulation is important for removing hydrogen bubbles from the surface of the electrodes and replenishing the electrolyte with fresh reactants. Improving the circulation of the electrolyte around the electrodes can help to remove hydrogen bubbles more efficiently, increasing the yield of hydrogen gas.
Fifth, Reduce impurities. Impurities in the electrolyte, such as dissolved salts or other substances, can interfere with the electrolysis reaction and reduce the efficiency of hydrogen bubble formation and collection. Reducing impurities in the electrolyte can help to improve the efficiency of the reaction, increasing the yield of hydrogen gas. Purification techniques such as distillation or reverse osmosis can be used to remove impurities from the electrolyte before electrolysis.
Hands-On Experience and Analysis
Here are my understanding during the hands-on experience on Rasendriya's house.
The hands-on experience covers the electrolysis procedure for capturing hydrogen gas, which involves the use of electrical energy to separate water molecules into hydrogen and oxygen gases. The method, commonly referred to as water electrolysis, involves running an electric current through a water solution that contains an electrolyte. When a current flows through water, the electrolyte, which aids in the conductance of electricity, causes the water molecules to disintegrate into gases of hydrogen and oxygen.
The cathode, the electrolysis cell's negatively charged electrode, collects hydrogen gas, whereas the anode, the cell's positively charged electrode, collects oxygen gas. To produce hydrogen gas, the electrolysis process necessitates a substantial amount of electricity, which must be kept in mind.
Electrolysis's effect on the environment relies on where the energy comes from that powers the procedure. The technique can be regarded as an eco-friendly and sustainable way to produce hydrogen gas if the electricity is generated using renewable energy sources like solar or wind power. However, the process might not be as environmentally favorable if non-renewable resources like coal or natural gas are employed to create the power.
In conclusion, the electrolysis method for capturing hydrogen gas involves using electrical energy to separate water molecules into hydrogen and oxygen gases. Even though the process uses a lot of energy, creating hydrogen gas using renewable electricity sources can make it sustainable.
Hands-on PROJECT PROGRAMMING
Mid Term Presentation, https://docs.google.com/presentation/d/1apiYvWqWk88WQoymlGqpQGcTJl9D3t4Dy7Bs1Q1Dagk/edit#slide=id.g22c66782de7_0_40