Difference between revisions of "User:PyrenAdam"
| (6 intermediate revisions by the same user not shown) | |||
| Line 27: | Line 27: | ||
Today we learned the basics on using OpenModellica to simulate Internal Combustion Engine System. From the simplest thing, like installing the library, to adding parts and adjusting parameters on the model. | Today we learned the basics on using OpenModellica to simulate Internal Combustion Engine System. From the simplest thing, like installing the library, to adding parts and adjusting parameters on the model. | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| Line 55: | Line 42: | ||
The lesson first starts off with 4 keywords: | The lesson first starts off with 4 keywords: | ||
| + | |||
1. Desalination | 1. Desalination | ||
| + | |||
2. Biomass | 2. Biomass | ||
| + | |||
3. Pyrolysis | 3. Pyrolysis | ||
| + | |||
4. Drier | 4. Drier | ||
| + | |||
These 4 are all interconnected in one way or another. | These 4 are all interconnected in one way or another. | ||
| Line 64: | Line 56: | ||
Pertamina and other gas industries are currently working towards supplying renewable energy. They turn to gasification, which originates from pyrolysis. Pyrolysis itself is the process of thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. A very simple example of this mentioned in a previous class is a flame that burns a piece of paper. | Pertamina and other gas industries are currently working towards supplying renewable energy. They turn to gasification, which originates from pyrolysis. Pyrolysis itself is the process of thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. A very simple example of this mentioned in a previous class is a flame that burns a piece of paper. | ||
| + | |||
| + | |||
| + | ---- | ||
| + | |||
| + | == Hydrolysis Project == | ||
| + | |||
| + | Electrolysis is a process that involves the use of electric current to drive a non-spontaneous chemical reaction. In electrolysis, an electrolyte (a substance that conducts electricity when dissolved in water or melted) is subjected to an electric current, causing the ions in the electrolyte to move towards the electrode of opposite charge. At the electrodes, chemical reactions take place, and the ions are either oxidized or reduced, depending on whether they are positively or negatively charged. The electrode at which oxidation occurs is called the anode, while the electrode at which reduction occurs is called the cathode. Electrolysis is used in various industrial and scientific applications, such as the production of metals, the purification of metals, the production of hydrogen and oxygen gases, the production of chemicals, and the electroplating of metals onto other materials. | ||
| + | |||
| + | Hydrolysis is a chemical reaction in which water is used to break down a larger molecule into smaller molecules. In hydrolysis, a water molecule is split into a hydrogen ion (H+) and a hydroxide ion (OH-), which can then react with the molecule being hydrolyzed. Hydrolysis reactions can occur in both organic and inorganic molecules, and they are often used to break down complex molecules into simpler ones. For example, the digestion of food in the human body involves hydrolysis reactions breaking down proteins, carbohydrates, and fats into their respective building blocks, such as amino acids, glucose, and fatty acids. Hydrolysis reactions can also be used in the synthesis of new molecules. For example, ester hydrolysis is used to produce carboxylic acids and alcohols from esters. In addition, hydrolysis reactions are used in the manufacture of a variety of products, such as soaps, detergents, and paper. | ||
| + | |||
| + | Water can be created when hydrogen and oxygen molecules combine, and this reaction is highly exothermic. Due to its low auto-ignition temperature, it has the potential to be an alternative fuel for vehicles in the future. While hydrogen is currently produced from natural gas, this process generates carbon dioxide as a byproduct, which cancels out the advantages of using hydrogen as a fuel alternative. However, "green" hydrogen, which is produced without creating any byproducts, is achievable. Electrolysis is one such method that can be used to produce hydrogen from water. | ||
| + | |||
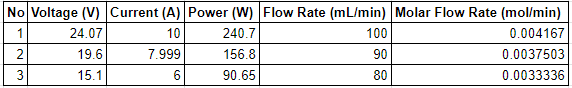
| + | An experiment was conducted with Rasen's equipment to study the effects of various metal plates as electrodes on the production of hydrogen. The variable changed is the current. The electrode used is one Aluminium 5000 series plate with a thickness of 5 mm. | ||
| + | [[File:Data_Project.png]] | ||
| + | |||
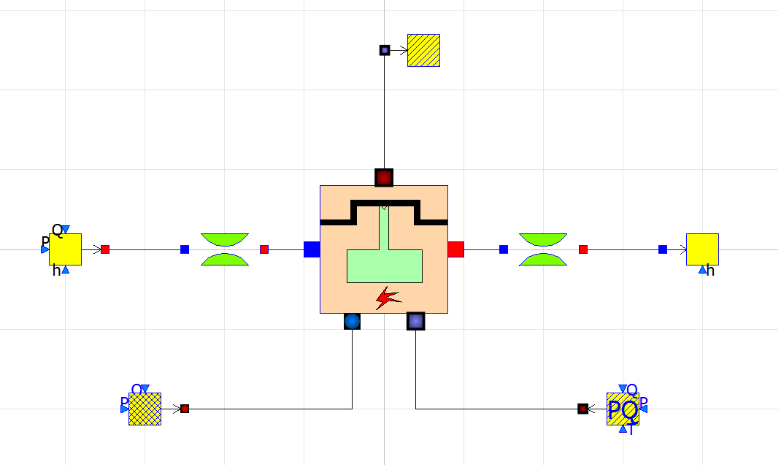
| + | Once the experiment has been conducted, the data can be used for simulation in OpenModellica. | ||
| + | |||
| + | [[File:Simulation.png]] | ||
Latest revision as of 07:14, 11 April 2023
Arfadhila Pyrenadi Adam
2006517764
Contents
My Chat with ChatGPT
How are sugarcane productions, battery, and fuel in relationship to one another?
ChatGPT:
Sugarcane production, battery, and fuel are related to one another through the production of biofuels, specifically ethanol. Sugarcane is a primary source of ethanol production, which can be used as fuel for vehicles or other applications.
The process of producing ethanol from sugarcane involves the fermentation of the plant's sugars, which are then distilled into ethanol. This ethanol can be blended with gasoline to produce a biofuel that can power vehicles.
In terms of batteries, sugarcane is also being explored as a potential source for producing high-performance lithium-ion batteries. Researchers have found that a material called lignin, which is present in sugarcane, can be used as a component in the cathodes of these batteries.
Overall, sugarcane has the potential to play an important role in the production of both biofuels and batteries, which are important for reducing our reliance on fossil fuels and transitioning towards more sustainable energy sources.
Intro to OpenModellica
Friday 24 February 2023
Today we learned the basics on using OpenModellica to simulate Internal Combustion Engine System. From the simplest thing, like installing the library, to adding parts and adjusting parameters on the model.
ECS 2 Class 28/02/2023
What actually happens during combustion is vibration. Everything around us is caused by vibration. When talking about any sort of environment, vibration is always related. Electrical appliances with displays are able to show images due to vibration of particles in the screen, releasing light of different frequency. A flame burns a piece of paper.
ECS 2 Class 07/03/2023
The lesson first starts off with 4 keywords:
1. Desalination
2. Biomass
3. Pyrolysis
4. Drier
These 4 are all interconnected in one way or another.
Desalination is used to produce drinking water from regular sea water. There are two main source of demands for drinking or clean water, the general public and the industries. Specifically, there is a program that is run by PLN for carbon capture that is somewhat connected to batteries. This program requires massive amount of resources, thus it should be done through joint effort by multiple entities. On the other hand, sea water is by no means clean. On a microscopic scale, filters made of graphene are used. Graphene is allotrope of carbon consisting of a single layer of atoms arranged in a hexagonal lattice nanostructure. The name is derived from "graphite" and the suffix -ene, reflecting the fact that the graphite allotrope of carbon contains numerous double bonds. On a macroscopic level, debris throughout the sea is plentiful. Efforts for cleaning this waste are very much mechanical. It takes massive resources, such as ships, to collect this waste. Even then, most waste collected originate from the surface.
Pertamina and other gas industries are currently working towards supplying renewable energy. They turn to gasification, which originates from pyrolysis. Pyrolysis itself is the process of thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. A very simple example of this mentioned in a previous class is a flame that burns a piece of paper.
Hydrolysis Project
Electrolysis is a process that involves the use of electric current to drive a non-spontaneous chemical reaction. In electrolysis, an electrolyte (a substance that conducts electricity when dissolved in water or melted) is subjected to an electric current, causing the ions in the electrolyte to move towards the electrode of opposite charge. At the electrodes, chemical reactions take place, and the ions are either oxidized or reduced, depending on whether they are positively or negatively charged. The electrode at which oxidation occurs is called the anode, while the electrode at which reduction occurs is called the cathode. Electrolysis is used in various industrial and scientific applications, such as the production of metals, the purification of metals, the production of hydrogen and oxygen gases, the production of chemicals, and the electroplating of metals onto other materials.
Hydrolysis is a chemical reaction in which water is used to break down a larger molecule into smaller molecules. In hydrolysis, a water molecule is split into a hydrogen ion (H+) and a hydroxide ion (OH-), which can then react with the molecule being hydrolyzed. Hydrolysis reactions can occur in both organic and inorganic molecules, and they are often used to break down complex molecules into simpler ones. For example, the digestion of food in the human body involves hydrolysis reactions breaking down proteins, carbohydrates, and fats into their respective building blocks, such as amino acids, glucose, and fatty acids. Hydrolysis reactions can also be used in the synthesis of new molecules. For example, ester hydrolysis is used to produce carboxylic acids and alcohols from esters. In addition, hydrolysis reactions are used in the manufacture of a variety of products, such as soaps, detergents, and paper.
Water can be created when hydrogen and oxygen molecules combine, and this reaction is highly exothermic. Due to its low auto-ignition temperature, it has the potential to be an alternative fuel for vehicles in the future. While hydrogen is currently produced from natural gas, this process generates carbon dioxide as a byproduct, which cancels out the advantages of using hydrogen as a fuel alternative. However, "green" hydrogen, which is produced without creating any byproducts, is achievable. Electrolysis is one such method that can be used to produce hydrogen from water.
An experiment was conducted with Rasen's equipment to study the effects of various metal plates as electrodes on the production of hydrogen. The variable changed is the current. The electrode used is one Aluminium 5000 series plate with a thickness of 5 mm.
Once the experiment has been conducted, the data can be used for simulation in OpenModellica.